WGSR - Pt/MgO
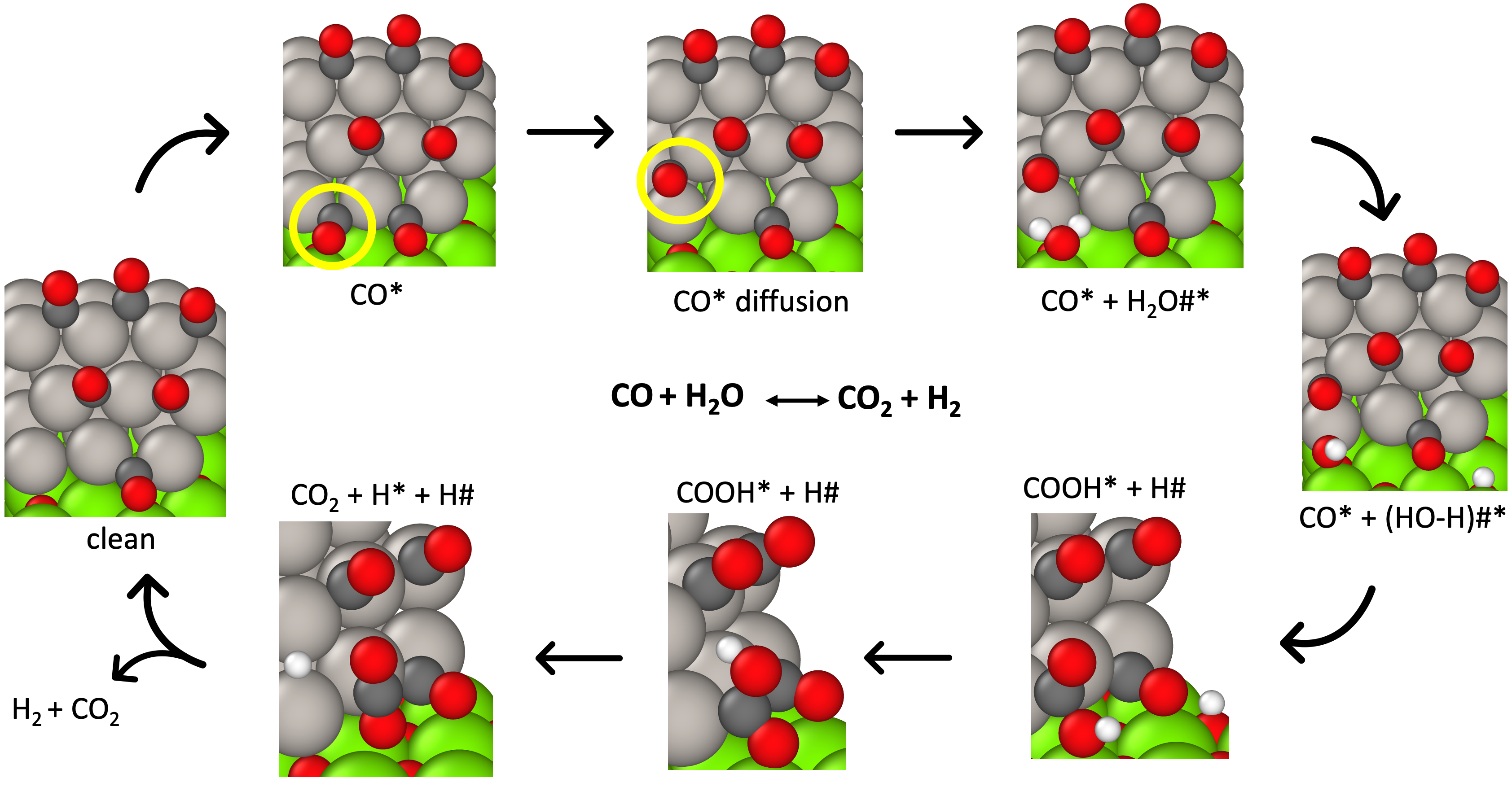
Catalysis at Metal/Oxide Interfaces - Water Gas Shift at Pt/MgO Boundaries
Catalysts comprising of metal nanoparticles dispersed on oxide supports have found applications in a vast number of chemical processes related to energy generation and environmental. In some cases, the interface between the metal and the supporting oxide is theorized to exhibit unique reactivity compared to just the metal or the oxide in isolation.
Water-Gas Shift reaction (WGSR) is one such reaction which is expected to show sensitivity to such metal/oxide interfaces. We investigate WGSR with Pt nanoparticles supported MgO as a model system, to understand how the reaction proceed at these interfacial sites.
Key takewaways:
-
The interfacial region greatly accelerate the water dissociation step which has an exceedingly high barrier on just the metal (Pt) or oxide (MgO) in isolation.
-
Partial poisoning of CO on the metal affects the over mechanism and it is important the model considers this effect explicitly.
-
When developing reaction network schemes for multi-components systems, the morphology of active site plays a crucial role with far-reaching consequences.